SAB BIO Presents Additional Clinical and Mechanistic Data from SAB-142 Phase 1 Trial in Adult Patients with Established Autoimmune Type 1 Diabetes at IDS 2026
All 4 T1D participants receiving SAB-142 showed C-peptide preservation with 3 of the 4 participants showing a super responder profile concomitant with T cell exhaustion
T1D participants receiving SAB-142 showed improved glycemic control
MIAMI, April 22, 2026 (GLOBE NEWSWIRE) -- SAB Biotherapeutics, Inc. (Nasdaq: SABS), a clinical-stage biopharmaceutical company developing a fully human anti-thymocyte immunoglobulin (hATG) for type 1 diabetes (T1D) and other autoimmune diseases, today presented additional clinical and mechanistic data from the Phase 1 HUman anti-thymocyte biologic in first-in-MAN (HUMAN) clinical trial of SAB-142 at the 21st Immunology of Diabetes Society (IDS) Congress in Brisbane, Australia.
The additional data presented for SAB-142 highlighted C-peptide preservation with a super responder profile among 3 of the 4 participants with T1D, correlated with evidence of CD4+ T conventional (Tconv) cell exhaustion. Other findings presented include improved glycemic control vs baseline based on continuous glucose monitoring (CGM) time in range data.
“These new results from the participants with T1D in the Phase 1 trial reinforce SAB-142’s intended mechanism of action, inducing T cell exhaustion that correlates with anticipated C-peptide response levels, and improved glycemic control not driven by exogenous insulin. These findings are consistent with what has been observed with rabbit ATG,” said Alexandra Kropotova, M.D., MBA, Chief Medical Officer of SAB BIO. “Importantly, our data demonstrated sustained immunomodulation without immunodepletion with both induction and maintenance dosing, a finding that directly supports SAB-142's differentiation as a potentially safe and effective long-term immunotherapy for patients across all stages of T1D.”
The Phase 1 T1D cohort included six adult participants (n=6), with four receiving SAB-142 at 2.5 mg/kg (n=4) and two receiving placebo (n=2). Participants ranged in age from 19 to 40 years. All participants with established T1D (Stage 3 T1D diagnosis within 28-40 months at the time of randomization) had residual beta cell function (C-peptide >0.2 nmol/L) and at least one T1D autoantibody at baseline. Phase 1 exploratory efficacy endpoints were measured at the End of Study Day 120 post SAB-142 administration. One placebo participant (n=1) completed through Day 120 as the other placebo participant discontinued early due to personal reasons.
T1D Cohort: C-Peptide Preservation and CD4+ Tconv Cell Exhaustion
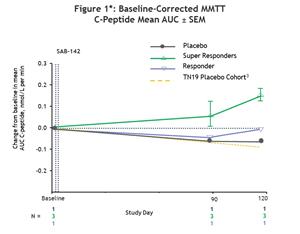
All participants treated with SAB-142 (2.5mg/kg) showed preservation in C-peptide levels compared to baseline, with 3 of the 4 participants exhibiting mean C-peptide values consistent with super responder status as previously categorized in the study of rabbit ATG (rATG).1 In that rATG study, super responders were defined as participants with C-peptide at or above the baseline at the end of the study (Month 24).1 Similarly, in the SAB-142 Phase 1 trial, participants with C-peptide response above the baseline at the end of the study (Day 120) were defined as super responders. Super responders demonstrated an increase in mean C-peptide values above baseline through Day 120, while the remaining treated participant showed a stabilized C-peptide level relative to baseline. In contrast, the placebo-treated participant showed a decline in C-peptide consistent with the predicted rate of disease progression.
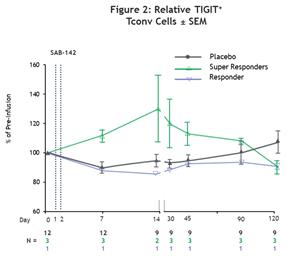
SAB-142 induced CD4+ Tconv cell exhaustion, as measured by TIGIT⁺ Tconv cell levels, contributing to C-peptide preservation, which is consistent with the mechanism of action observed with rATG in previous studies.1,2 Super responders demonstrated an early and sustained increase in TIGIT⁺ Tconv cell levels post-infusion. In contrast, the placebo-treated participants, including both healthy volunteer (HV) and T1D participants, remained near baseline through the study period.
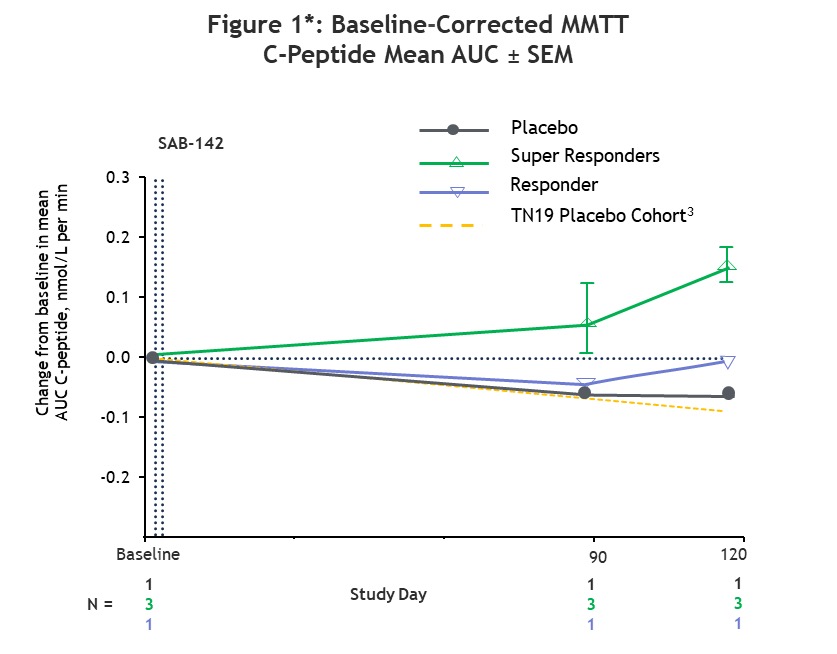
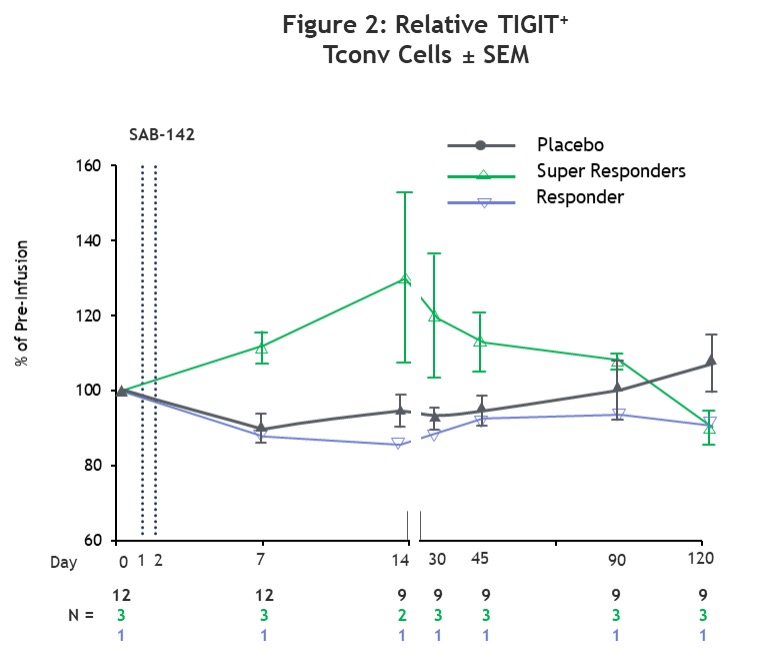
*Note: 3Predicted decline was estimated by first calculating the incremental AUC of TN19 placebo subject C-peptide MMTT data (n=26) for Weeks 48-96 with post-2hr values masked. The linear slope was used to calculate the predicted rate of decline in days: AUCBL – (-0.6108*(Study Day/7)). Source: Haller et al. Diabetes. 2019 Jun;68(6):1267–1276.
MMTT = Mixed Meal Tolerance Test; AUC = Area Under the Curve; SEM = Standard Error of the Mean.
T1D Cohort: Glycemic Control Improvement based on CGM Time in Range (TIR)
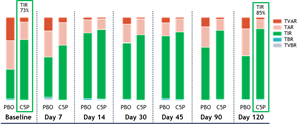
Participants receiving SAB-142 (2.5 mg/kg) demonstrated an increase in mean TIR from 73% at baseline to 85% at Day 120, meaning SAB-142 effects on glycemic control are consistent with C-peptide response. Additionally, the improvement in glycemic control was not associated with increases in exogenous insulin use. The Phase 1 study participants are patients with established T1D who are experienced in managing their disease for up to 4 years and are beyond their T1D “honeymoon” period.
Figure 3: CGM Mean Time in Range ± SEM
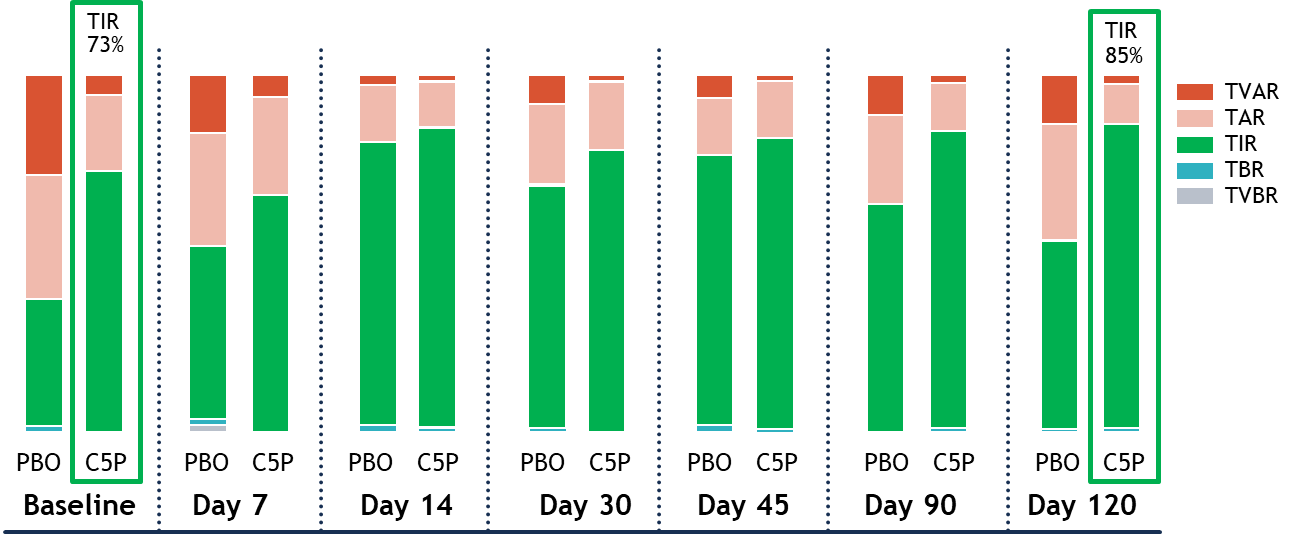
TVAR = Time Very Above Range; TAR = Time Above Range; TIR = Time in Range; TBR = Time Below Range; TVBR = Time Very Below Range.
“We were excited to present these new findings to the global scientific community at the Immunology of Diabetes Society Congress, as they provided deeper insights into how SAB-142’s immunologic effects may translate into meaningful benefit for people with T1D,” said Samuel J. Reich, Chief Executive Officer of SAB BIO. “In sharing these Phase 1 data, we continue to build confidence in SAB-142’s differentiated and potentially best-in-class product profile as a disease-modifying therapy for T1D. We look forward to reporting topline data from our registrational Phase 2b SAFEGUARD trial in the 2H 2027.”
Full data presented at IDS 2026 is now available to review in the Presentations section of the Company’s website.
About the Phase 1 HUMAN Trial of SAB-142
The Phase 1 HUman anti-thymocyte biologic in first-in-MAN (HUMAN) clinical trial of SAB-142 is a randomized, double-blind, placebo-controlled, single-ascending dose and redose, adaptive design clinical study among healthy volunteers and one cohort of adult participants with established T1D. The study objectives include establishing safety, tolerability, pharmacokinetic (PK), immunogenicity, and pharmacodynamic (PD) profile for SAB-142 with a single 0.03-4.5mg/kg dose plus one cohort with an additional 1.5mg/kg dose.
About the SAFEGUARD Trial
SAFety and Efficacy of human anti-thymocyte immunoGlobUlin SAB-142 ARresting progression of type 1 Diabetes (SAFEGUARD) trial is a randomized, double-blind, placebo-controlled multi-center Phase 2b study designed to assess the safety, efficacy, and tolerability of SAB-142 in patients with new onset Stage 3 T1D. The SAFEGUARD trial is actively enrolling and dosing participants at multiple sites around the world. SAB-142 is in development as a novel, potentially best-in-class, disease-modifying immunotherapeutic approach to treat T1D by delaying the progression of disease. SAFEGUARD Part A is a dose-ranging study in adult patients. SAFEGUARD Part B is a randomized double-blind, placebo-controlled, dose-ranging study. Enrolled patients will receive two SAB-142 or placebo infusions six months apart. All patients, including the placebo-control group, are eligible for the 12-month long-term extension study upon study completion. Additional details are available on www.clinicaltrials.gov (NCT07187531) and at https://safeguardstudy.com/.
About SAB-142
SAB-142 is a potentially disease-modifying, redosable immunotherapy in clinical development for the treatment of autoimmune type 1 diabetes (T1D). SAB-142 is a multi-specific, fully human anti-thymocyte globulin (hATG) with a mechanism of action analogous to that of rabbit ATG (rATG). rATG has demonstrated in multiple clinical trials the ability to slow disease progression in patients with new- or recent-onset of Stage 3 T1D. SAB-142, like rATG, directly targets multiple immune cells involved in destroying pancreatic beta cells, including modulation of “bad acting” T-lymphocytes. By stopping immune cells from attacking beta cells, this treatment has the potential to preserve insulin-producing beta cells.
About SAB BIO
SAB BIO is a clinical-stage biopharmaceutical company focused on developing multi-specific, high-potency, human immunoglobulin G (hIgG) to treat and prevent immune and autoimmune disorders. Using advanced genetic engineering and antibody science, SAB BIO developed a proprietary technology which holds the potential to generate additional novel therapeutic candidates utilizing the human immune response, without the need for human donors or convalescent plasma. SAB BIO has optimized genetic engineering in the development of transchromosomic cattle, or Tc-Bovine, to produce hIgG. SAB BIO’s drug development production system is able to generate a diverse repertoire of specifically targeted, high-potency, hIgGs that can address a wide range of serious unmet needs in human diseases. The Company’s lead candidate, SAB-142, targets autoimmune T1D with a disease-modifying therapeutic approach that aims to change the T1D treatment paradigm by delaying onset and potentially preventing disease progression of Stage 3 T1D patients. SAB-142 is currently being evaluated in newly diagnosed Stage 3 autoimmune T1D patients in a registrational Phase 2b clinical trial called SAFEGUARD. For more information, visit www.sab.bio.
Forward-Looking Statements
Certain statements made in this press release that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “to be,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook,” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding future events, including statements about the development and clinical trial results of the Company’s T1D program and other discovery programs.
These statements are based on the current expectations of SAB BIO and are not predictions of actual performance, and are not intended to serve as, and must not be relied on, by any investor as a guarantee, prediction, definitive statement, or an assurance, of fact or probability. These statements are only current predictions or expectations, and are subject to known and unknown risks, uncertainties and other factors which may be beyond our control. Actual events and circumstances are difficult or impossible to predict, and these risks and uncertainties may cause our or our industry’s results, performance, or achievements to be materially different from those anticipated by these forward-looking statements. A further description of risks and uncertainties can be found in the sections captioned “Risk Factors” in our most recent annual report on Form 10-K, subsequent quarterly reports on Form 10-Q, as may be amended or supplemented from time to time, and other filings with or submissions to, the U.S. Securities and Exchange Commission, which are available at https://www.sec.gov/. Except as otherwise required by law, SAB BIO disclaims any intention or obligation to update or revise any forward-looking statements, which speak only as of the date they were made, whether as a result of new information, future events, or circumstances or otherwise.
________________
References:
References to third-party publications are provided for informational purposes only.
1Haller MJ, Gitelman SE, Gottlieb PA, et al. Antithymocyte Globulin Plus G-CSF Combination Therapy Leads to Sustained Immunomodulatory and Metabolic Effects in a Subset of Responders with Established Type 1 Diabetes. Diabetes. 2016;65(12):3765-3775. doi: 10.2337/db16-0823
2Jacobsen LM, Diggins K, Blanchfield L, et al. Responders to Low-Dose ATG Induce CD4+ T Cell Exhaustion in Type 1 Diabetes. JCI Insight. 2023;8(16):e161812. doi: 10.1172/jci.insight.161812
3 Haller MJ, Long SA, Blanchfield JL, et al; Type 1 Diabetes TrialNet ATG-GCSF Study Group. Low-Dose Anti-Thymocyte Globulin Preserves C-Peptide, Reduces HbA1c, and Increases Regulatory to Conventional T-Cell Ratios in New-Onset Type 1 Diabetes: Two-Year Clinical Trial Data. Diabetes. 2019;68(6):1267-1276. doi:10.2337/db19-0057
CONTACTS
Investor Relations:
Christine Ryan
ir@sab.bio
Media:
Sheila Carlson
media@sab.bio
Figures accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/2c1f3e53-a4ae-44fe-8244-e1c6273f067a
https://www.globenewswire.com/NewsRoom/AttachmentNg/6beeab49-fb1f-4f9f-a34d-35958c65fd7a
https://www.globenewswire.com/NewsRoom/AttachmentNg/7d3ea041-0c84-4609-b7db-3218e361bdb2

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.